



A new composition-dependent equation of state for clinopyroxene
Simon Schorn & Eleanor C.R. Green
Session 07a: Thermodynamics and Kinetics of Mineral-Fluid-Melt Interactions in the Global Element Cycling
Introduction
Clinopyroxene (cpx) is a main rock-forming mineral in basic- and intermediate lithologies, as well as in the mantle of planetary bodies. Because it can assume a range in compositions, it comes in many different shapes and colors (see Figures). Its incredible variability poses a serious challenge when it comes to understand the thermodynamics of clinopyroxene.
Thermodynamic modelling & cpx
Thermodynamic modelling has become a staple in the petrologist’s toolbox – to simulate igneous, metamorphic and geodynamic processes shaping the Earth and other rocky planets such as Mars. To do this we require realistic descriptions of the minerals that make up the rocks we want to investigate – so-called composition-dependent equations of state (x-eos). These are used for calculations with phase equilibrium software packages such as THERMOCALC and Perple_X.
So far several x-eos describing clinopyroxene exist, but they each are designed to work for specific conditions of pressure–temperature–composition (P–T–x). Unfortunately, these x-eos are not consistent with each other, and cannot be used to model processes that encompass more than one of these regimes. We therefore need a ‘single’ model that envelopes all (or most) possible varieties and flavors of cpx occurring in nature.
The solution: Cpx-Wing
We are trying to combine the strengths of the pre-existing x-eos into one monster-model – that we affectionately call Cpx-Wing:
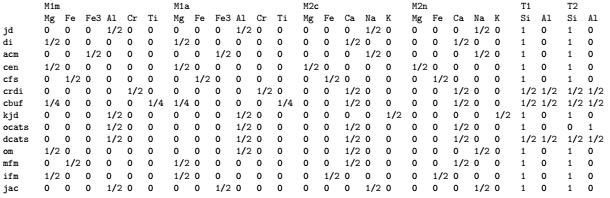
Cpx has two octahedral crystallographic sites, M1 & M2, and one tetrahedral site (T) that can accommodate various cations. To simulate natural cpx where cations occupy sites in an ordered manner, the sites need to be split, as shown above. The abbreviations in the list are the compositional end-members that are required to build Cpx-wing. Because cpx can assume a wide range of compositions, a great number of end-members are required, which is why the above list looks as it does.
So does this super-model work already?
Yes, but no. We have successfully combined the pre-existing x-eos into a ‘working’ model that successfully calculates cpx-bearing equilibria, but we still need some fine-tuning of the calibration in order to simulate realistic rocks. For this we use experimental data that we try to fit as close as possible.
Problems & pitfalls
A major problem when designing x-eos is that the minerals making up the systems we are trying to simulate are extremely complex and involve several mechanisms that are poorly understood. Basic rocks in particular typically consist of very few minerals, for example the eclogite shown in the Figure on the top left. It essentially contains only omphacitic clinopyroxene and garnet, but in those two minerals all of the rock’s chemical components are accommodated. This means that there are countless possible combinations of compositional partitioning between the two minerals. This makes them very difficult to constrain.
Another issue is that basic rocks contain other minerals that show a wide compositional variability that makes them equally poorly constrained, for example amphiboles (e.g. the dark green hornblende in the granulite from Namaqualand above). Because these minerals can be dominant in models of basic rocks, their behaviour majorly influences the composition of other coexisting phases, such as cpx.
We are therefore working hard to reconcile the delicate interactions of clinopyroxene and amphiboles in our new x-eos, using both high-P–high-T experimental data and natural equilibria. We are aiming to add Cpx-wing to your thermodynamic arsenal later in the year.
Thank you very much for viewing our work!
Check in for some Q&A with Simon on Tuesday 23rd June between 11:00–12:00 and 19:45–20:45 (HST).
Please visit our website and follow us on twitter.



