The HPx-eos and THERMOCALC program can be described as tools for use within the field of computational phase equilibrium modelling. Below we give a broad overview of the field. See also our Resource hub.
An introduction to phase equilibrium modelling
How it works
The study of equilibrium phase relations underpins our understanding of igneous and metamorphic rocks – those formed by crystallisation from a magma, or recrystallisation of an existing mineral assemblage. Such (re-)crystallisation is driven by the need of the rock or rock/fluid system to evolve towards thermodynamic equilibrium, or try to maintain its proximity to equilibrium as the physical conditions change. We therefore make numerical models of equilibrium phase relations and compare these with the phase assemblages found in rocks, hoping to gain insight into Earth processes. In the discussion below, we make the commonplace simplification that rocks evolve at superimposed pressure (P) and temperature (T) and a constant or systematically evolving bulk composition.
Thermobarometry
In a typical use of phase equilibrium modelling, compositional analyses of a rock are compared with computed phase relations to infer the P and T at which the rock was last in equilibrium – the point at which it ceased to evolve because kinetic barriers to equilibration became prohibitive. In this paradigm, the mineral assemblage and mineral compositions represent a snapshot in the rock’s history, which might be the cooling history of a cumulate pile, or the burial and exhumation of a metamorphic rock.

The estimate of such a short-lived (P, T) coordinate is thermobarometry. It may involve a direct inversion of the analysed compositions of minerals believed to be in equilibrium. Such an approach has, of course, a long history. In classical thermobarometry, individual equilibrium equations are calibrated to give a value for either P or T, rather than modelling full compositionally-dependent equations of state (x-eos) for the phases involved. Throughout this website, the inverse method we discuss is multiple-reaction thermobarometry, which does use x-eos, and obtains average estimates of P and/or T based on an independent set of all the available equilibrium equations that both modelling and analysis make accessible. The averaging procedure accounts for correlated uncertainties in the modelling and analyses.
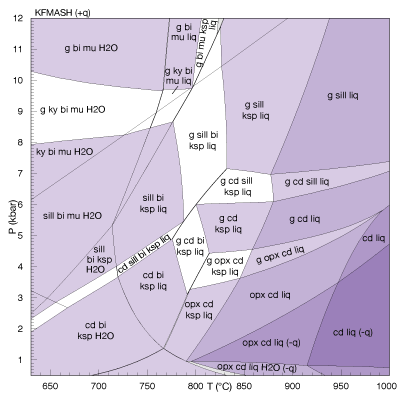
In metamorphic geology these days, a common alternative to inverse thermobarometry is pseudosection thermobarometry: the forward modelling of phase relations at a constrained bulk composition, representing that of the equilibrium assemblage inferred for the rock. This approach uses x-eos, as for multiple-reaction thermobarometry. A map or pseudosection of the most stable phase assemblages seen by the bulk composition is calculated, and compared with the observed assemblage. If the observed assemblage can be matched with a calculated field, the position and nature of the field in P–T space gives an estimate of the equilibrium (P, T) coordinate and its uncertainty.
Open-system processes
Of course, many Earth processes involve systems that are continuously or episodically open to chemical change. In metamorphic geology, aqueous fluid or melt might be lost when a threshold porosity is exceeded, or may inflitrate from elsewhere. The centres of growing porphyroblasts may become progressively isolated from the surrounding matrix. Many problems in igneous geology are concerned with fractional crystallisation and the subsequent isolation of crystals from the parental magma, perhaps punctuated by injections of new magma. Such scenarios can be forward-modelled as repeated calculations of equilibrium phase relations, subject to a changing bulk composition of the system.
The modelling of processes implies a temporal dimension to the problem, while changes in bulk composition imply a spatial dimension, with mass moving from one place to another while undergoing chemical fractionation. Clearly, there is an opportunity to integrate such calculations with geodynamics simulations, based on a formal timestep and spatial grid. Feedback then takes place between the phase equilibrium calculations (providing assemblages and their thermodynamic properties) and geodynamics (providing the physical response to chemical changes, which in turn drives further chemical changes). We are currently exploring this with our collaborators from the Geophysics and Geodynamics Group at the University of Mainz and the Australia-based Underworld team.
Role of the HPx-eos and THERMOCALC
The building blocks of our phase equilibrium modelling work are the HPx-eos, incorporating end-members from the Holland & Powell (2011) dataset. Each HPx-eos represents the thermodynamics of one phase – a mineral, aqueous fluid or silicate melt. Used together and suitably manipulated, they generate a set of model phase relations that can be compared with rock samples or used in simulations of Earth processes. The definitive implementation of the HPx-eos is found in THERMOCALC, our phase equilibrium calculation program.
Data and internal consistency
Calibration of the HPx-eos involves data drawn directly from natural and experimental phase relations, combined with a vastly larger body of experimental data on the calorimetric and volumetric properties of mineral end-members. The latter is incorporated via the Holland & Powell (2011) dataset. We aim for internal consistency in our approach to modelling phase equilibria, combining data from disparate sources by optimisation within a mathematical framework suggested by thermodynamic theory. More details can be found in our discussion of How the HPx-eos work (see also our footnote on credit for experimental work).
Getting started
We suggest the following as prerequisites for computational phase equilibrium modelling:
- Excellent petrography and analysis, representing a system meaningfully close to equilibrium.
- A comprehensive understanding of what insight phase equilibrium modelling could in principle bring to the observations, with an understanding of the uncertainties and limitations involved. Our Resource hub may help.
The modeller must then:
- decide which thermodynamic models for phases/phase equilibria to use. Our models, the HPx-eos, can be applied in a number of crustal and upper mantle contexts. There are many other possibilities.
- decide which software to use – it must be compatible with the chosen models.
- consider uncertainties in the modelling.
- remain professionally sceptical! When results are obtained, consider if they make sense. If they don’t, it could mean one of the following, for which the Resource hub may provide help:
- the problem is not being modelled in an appropriate way – often the bulk composition used in calculations is not close enough to the bulk composition at which the equilibrium assemblage developed, or perhaps chemical potential or volume would be better independent variables than bulk composition or pressure
- the problem is not suitable for phase equilibrium modelling at all – probably because the equilibrium assumption doesn’t hold on a useful lengthscale here
- you are expecting too much, given the uncertainty in the modelling, or the textural complexity of the rock
- the x-eos are not behaving well. In this case the developers will be pleased to hear about it so they can improve future calibrations (contact us about the HPx-eos!).
References and footnotes
Holland & Powell (2011) J Metamorph Geol 29 333-383.
The concept of a phase diagram at constrained bulk composition is thanks to Bas Hensen. This diagram is given various names in the literature. Here we use the term pseudosection to refer to such diagrams, plotted in P-T, P-X or T-X space. The term pseudosection is intended in the sense of a pseudobinary – the compositions of the phases lie in general outside the plane of the section.
